October 3, 2022
MorphOMICs: Revealing the Hidden Meaning of Microglia Shape
Novel neuroscientific method reveals correlations between shapes of microglia cells
Grasping the complexity of the brain is one of the major quests in science. To understand its function and adaptation in disorders, neuroscientists turn to microglia cells. Now, an international team of scientists from the Siegert group at the Institute of Science and Technology, Austria have developed MorphOMICs: a pioneering data-driven method that unfolds a map of how brain region, sex, and disease progression influence microglia – all by looking at their shape.

Microglia cells are the brain’s canary in the coal mine. In the olden times, miners took canary songbirds into the mine with them, as they are much more sensitive than humans to the deadly carbon monoxide found underground. The birds reacted first, issuing a warning to the miners. Analogously, whatever happens to a brain, the microglia are the first to respond. They are involved in Alzheimer’s disease, brain development disorders, migraine, and even psychological disorders. Yet, how we can detect their response early on, before the onset of neurological symptoms is a pressing issue of ongoing research in neuroscience.
“Once you see microglia, you are fascinated by how strange they look!” A trained statistical physicist Ryan John Cubero, a Postdoc in Sandra Siegert’s group at the Institute of Science and Technology Austria (ISTA) explains why: “The shape of microglia carries information about what they are doing. The way they look – their morphology – is basically a signature of their function. Until now, no suitable method existed to study this!”
In fact, the common method to investigate microglia morphology, called morphometrics, is insufficient. “Classically, microglia are distinguished into two functional states: the surveillant state, where it screens the local environment if something is ‘off’, and the activated state, where it eats up bad stuff in a process called phagocytosis,” explains neuroscientist and doctoral student Gloria Colombo, first author of a recent paper published in Nature Neuroscience. “But in reality, these two are only extreme states on a wide spectrum of intermediate ones, which are not captured with traditional morphometrics. Our method closes this important gap.” With MorphOMICs, the team provides a powerful tool to address the functional state of microglia. Their publication in Nature Neuroscience also puts MorphOMICs to practice. On employing it, they found that microglia differ in adult male and female mouse brains. These differences are even more pronounced during early development and again during Alzheimer’s disease.
A cross-disciplinary achievement
From, previous work the scientists knew that ketamine—an anesthetic drug—has dramatic effects on the brain. Microglia suddenly starts to remove a structure that stabilizes the neural connections of the brain, thus, returning it to a state of high plasticity as seen in younger brains. “At first, when we looked at the shapes of the microglia, we couldn´t resolve morphological differences. This was a real struggle,” reports Assistant Professor Sandra Siegert. “We got inspired to look at the topology of the cells in a lecture by mathematician Kathryn Hess during the ISTA colloquium series. This started to be fruitful and inspiring cooperation across disciplines.”
Algebraic topology analyzes the properties of geometric objects. The cells’ intricate tree-like structure, examined from a topological point of view, reveals previously hidden information. “Truly, we were shocked at how remarkably this worked – seeing how microglia morphologies unfold for each brain region, sex, and how they change during disease!”
MorphOMICs allowed the authors to build an atlas of microglia morphologies. A certain shape of microglia is one location on the map. Neighboring locations on the map are microglia that are similar in terms of their topology. To understand this, imagine trees: Two trees with different lengths and directions of branches may have the same abstract way of forking. Therefore, they would occupy the same location on the map. “As microglia are very active, we couldn´t apply topology straightaway. We first had to validate the statistical approach with our dataset of more than 40,000 microglia cells, and then we could use machine learning strategies to visualize the atlas,” says Cubero, who has a background in statistical physics and has been responsible for the data science part of the project. “The atlas shows all possible microglia topologies. Invisible to the naked eye, we now see that certain brain regions exhibit specialized microglia topologies.”
Sex-dependent Alzheimer’s disease
Once the method was established, the scientists applied it. Together with collaborators at the Massachusetts Institute of Technology (MIT), they obtained mice that genetically reproduce Alzheimer’s disease symptoms. “Curiously, the developmental path for Alzheimer’s shows striking differences for the two sexes. Female microglia jump to an extreme state much quicker,” summarizes Colombo. “This hints at microglia morphology reacting much faster in females than in males for this degeneration.”
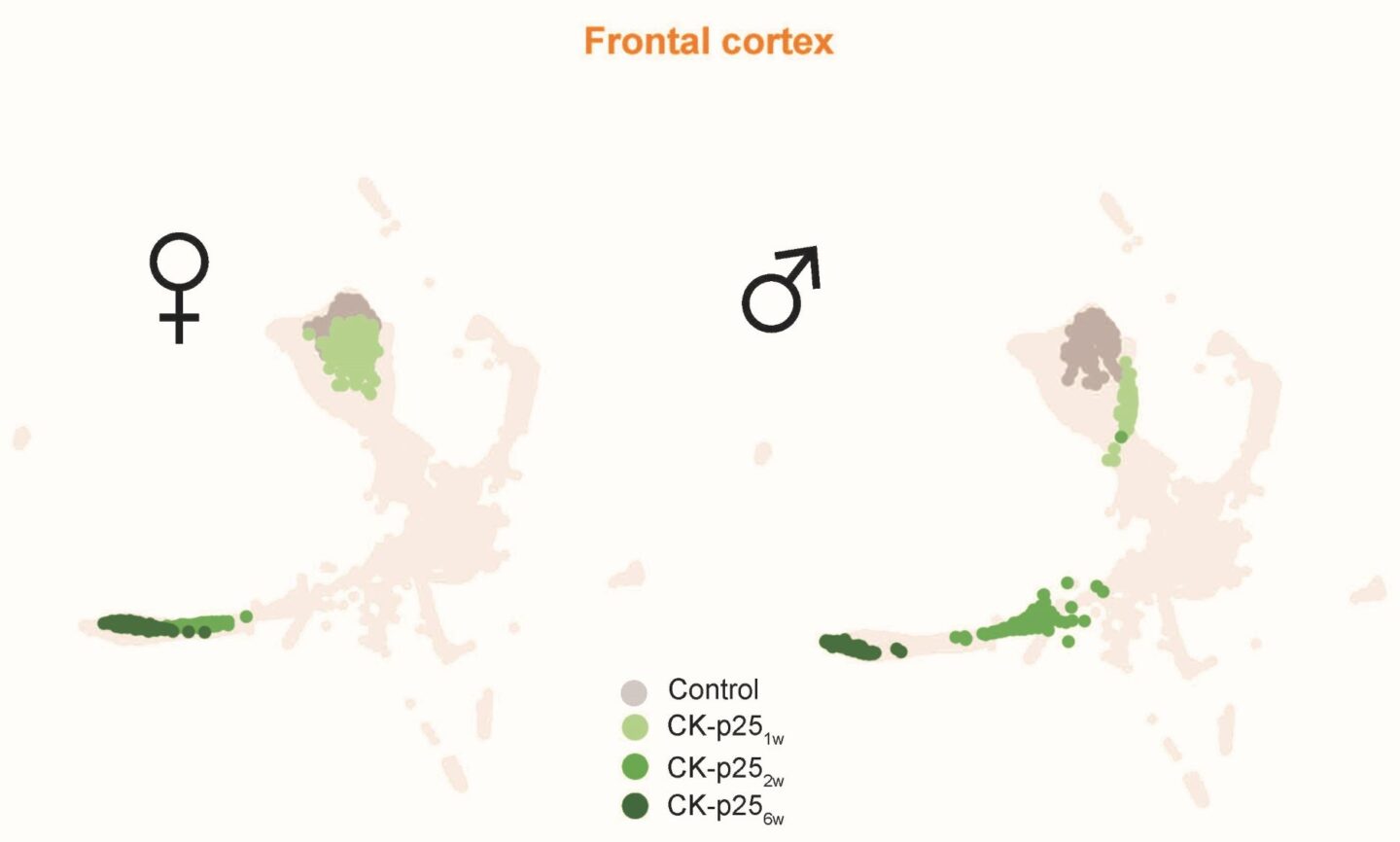
Legend for Image 3: Control: Adult healthy mouse, CK-p25: genetic mouse model of de-novo Alzheimer’s disease one- (1w), two- (2w), and 6-weeks (6w) after the induction of the degeneration
MorphOMICs can estimate the location of the respective microglia of a new condition on the map. The team applied this to the aforementioned study on ketamine anesthesia and could update their previous findings: General anesthesia is assumed to be a completely reversible process, but the topology of microglia uncovered that this is not the case. Microglia morphology does not recover to its initial condition. “What does this mean? Well, the mechanism we don´t know yet,” Cubero admits, “but this is only one example of how MorphOMICs support discoveries.”
In subsequent work, the team aims at resolving how microglial function relates to its morphology. Also, the method could be extended to all microglia, meaning it applies to all species including humans. “Of course, results from mice brains cannot be 1:1 translated to humans – but they provide first critical insights about what to look at,” says Sandra Siegert.
Publication:
Gloria Colombo, Ryan John A. Cubero, Lida Kanari, Alessandro Venturino, Rouven Schulz, Martina Scolamiero, Jens Agerberg, Hansruedi Mathys, Li-Huei Tsai, Wojciech Chacholski, Kathryn Hess, and Sandra Siegert. 2022. Microglial MorphOMICs unravel region- and sex-dependent morphological phenotypes from postnatal development to degeneration. Nature Neuroscience.
DOI: https://doi.org/10.1038/s41593-022-01167-6
Funding information:
The project members at ISTA were supported by the European Union Horizon 2020 research and innovation program under the Marie Skłodowska-Curie Actions program No.754411 and by the European Research Council (ERC) grant No. 715571.
Information on animal studies: The analysis of microglia morphologies is only possible by working with living animals since microglia change entirely in their genetic, molecular, and functional signature outside their brain environment. No other methods, such as in silico models, can serve as alternatives. The animals are raised and treated according to the strict regulations of Austrian law. All animal procedures at ISTA are approved by the Federal Ministry of Education, Science, and Research.



